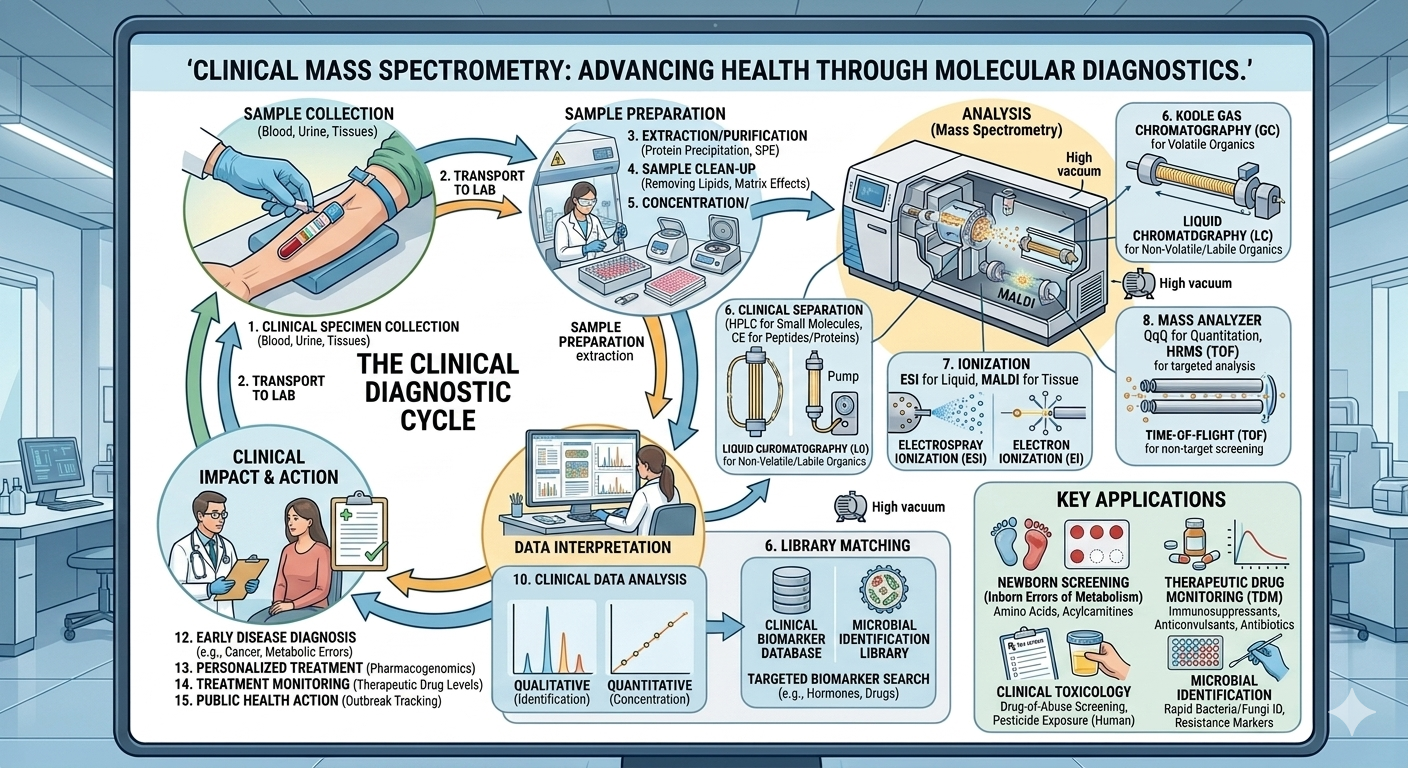
The global clinical mass spectrometry (MS) market is entering a high-growth phase, driven by the convergence of precision medicine, regulatory rigor, and the need for faster, in-house diagnostics. Valued at USD 1.92 billion in 2025, the market is projected to reach USD 3.31 billion by 2031, expanding at a CAGR of 9.5%. For laboratory leaders, hospital administrators, and diagnostics companies, this growth reflects a structural shift toward high-specificity, multiplexed testing platforms.
Why Clinical Mass Spectrometry Is Becoming Core to Modern Labs
Clinical laboratories are under increasing pressure to deliver accurate, rapid, and cost-efficient results while complying with stringent accreditation frameworks. Mass spectrometry—especially LC-MS/MS—addresses these demands by enabling:
- Ultra-low detection limits for small molecules
- High specificity compared to traditional immunoassays
- Multiplexing capability, allowing multiple analytes in a single run
- Reduced reliance on send-out testing, improving turnaround time (TAT)
This combination is making MS platforms indispensable in tertiary care hospitals, reference labs, and academic medical centers.
Download PDF Brochure @ https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=234108367
LC-MS Dominance: The Backbone of Clinical Workflows
Among techniques, liquid chromatography–mass spectrometry (LC-MS) leads the market and accounted for the largest revenue share in 2025. Its dominance is not incidental—it is rooted in operational and clinical advantages:
- Supports routine, reimbursable diagnostic tests
- Enables test menu consolidation on a single platform
- Improves workflow efficiency and lab economics
- Facilitates in-house testing of high-value assays
Key Use Cases Driving LC-MS Adoption
- Immunosuppressant drug monitoring
- Steroid hormone profiling
- Vitamin and metabolic panels
- Pain management and toxicology screening
Hospitals are increasingly investing in LC-MS systems to internalize testing, reducing outsourcing costs and gaining tighter control over quality and compliance.
Therapeutic Drug Monitoring (TDM): The Largest Application Segment
Therapeutic drug monitoring (TDM) remains the leading application area in the clinical MS market. Its importance is amplified by the rise in complex, multi-drug treatment regimens, particularly in:
- Oncology
- Transplant medicine
- Neurology (antiepileptics)
- Infectious diseases
LC-MS/MS has become the reference method for TDM due to:
- Superior analytical specificity
- Lower limits of detection
- Ability to quantify multiple drugs/metabolites simultaneously
These capabilities translate into better dose optimization, reduced adverse drug events, and improved patient outcomes—key priorities for value-based healthcare systems.
Component-Level Innovation Driving System Performance
The performance of clinical MS systems depends on advancements across core components:
- Mass analyzers: Enhancing resolution and accuracy
- Detectors: Improving sensitivity and signal detection
- Ion sources: Enabling stable ionization across diverse compounds
- Vacuum systems: Ensuring consistent instrument performance
Vendors are focusing on automation-ready, compact, and user-friendly systems to reduce dependency on highly specialized operators—one of the historical barriers to adoption.
Installed Base Expansion & Replacement Trends
The market is also being shaped by installed base growth and system replacement cycles:
- Aging instruments are being replaced with next-gen LC-MS/MS platforms
- Labs are upgrading to systems with integrated automation and software-driven workflows
- Increasing preference for IVD-validated assay kits over laboratory-developed tests (LDTs) in certain regions
This creates recurring revenue opportunities for manufacturers through service contracts, consumables, and software upgrades.
Regional Insights: Why the U.S. Leads North America
The United States is expected to maintain the largest share of the North American clinical MS market through 2031. Key structural advantages include:
- A high concentration of tertiary care hospitals and reference laboratories
- Strong adoption of high-complexity diagnostic testing
- Well-established reimbursement frameworks
- Supportive regulatory environment for advanced assays and LDTs
This ecosystem sustains both high instrument utilization rates and continuous test menu expansion, reinforcing market leadership.
Strategic Outlook: What Industry Leaders Should Watch
For stakeholders across diagnostics, medtech, and healthcare delivery, several strategic themes are emerging:
1. Shift Toward Fully Automated MS Workflows
Automation—from sample prep to data interpretation—will be critical for scaling adoption beyond specialized labs.
2. Integration with Clinical Informatics
MS platforms increasingly integrate with LIS and hospital IT systems, enabling real-time decision support.
3. Expansion of Clinical Applications
Beyond TDM and toxicology, growth is expected in:
- Endocrinology
- Metabolomics
- Infectious disease diagnostics
4. Regulatory and Standardization Push
Demand for IVD-certified assays and standardized workflows will shape purchasing decisions, particularly in regulated markets.
Conclusion
The clinical mass spectrometry market is transitioning from a specialized analytical tool to a mainstream clinical diagnostics platform. With LC-MS at the forefront and TDM driving test volumes, the market’s trajectory reflects a broader shift toward precision, efficiency, and in-house diagnostic control.
Organizations that invest in automation-ready systems, scalable workflows, and expanded test menus will be best positioned to capitalize on this growth curve through 2031.
Enquire Before Buying @ https://www.marketsandmarkets.com/Enquiry_Before_BuyingNew.asp?id=234108367
